Drs. Lorraine and Suneil Kalia from Krembil Brain Institute study Parkinson’s disease, from understanding the molecular biology to treating patients with medications and surgical therapies. (Photo: UHN)
A Phase I study co-led by UHN and Sunnybrook Health Sciences Centre is using focused ultrasound for the first time to open the blood-brain barrier and deliver a drug to a targeted part of the brain in patients with Parkinson’s disease.
Though cautiously optimistic, researchers hope this early research could one day pave the way to new treatments and, hopefully, a cure for Parkinson’s.
The world-class movement disorders team within the Edmond J. Safra Program in Parkinson’s Disease and the Morton and Gloria Shulman Movement Disorders Clinic at the Krembil Brain Institute, including Drs. Lorraine and Suneil Kalia, study Parkinson’s from understanding the molecular biology of the disease all the way to treating people diagnosed with the neurological disorder, both with medications, as well as surgical therapies.
Q-and-A with Dr. Lorraine Kalia, co-Principal Investigator, neurologist and Krembil Brain Institute clinician-scientist. Watch a video of Dr. Lorraine Kalia.
What makes this study unique?
This is a true collaboration. Dr. Nir Lipsman from Sunnybrook brings the expertise in focused ultrasound, and Suneil and I bring expertise in Parkinson’s research and treatment. As a neurologist, I have been treating and studying Parkinson’s disease over the last 10 years and in that time, I, and others have been challenged by not having any treatments that can actually stop or slow the progression of the disease. One of the major barriers is that, even if we had a drug or some sort of compound that could do that, it is very difficult to get it into the brain, particularly into the right area that needs to be treated. So, what makes this study unique is, we’re using focused ultrasound as a means to open up this barrier that keeps compounds or drugs from getting into the brain and we are using it in a very targeted fashion for the first time ever in people who have Parkinson’s disease.
What is the blood-brain barrier and why is it difficult for drugs to pass through it?
The blood-brain barrier is a collection of cells that protects the brain. It acts as a border to prevent compounds or harmful chemicals from getting into the brain, because the brain is an important organ. One of the things that the blood-brain barrier keeps from getting into the brain are particularly large molecules and so there are a many potential drugs that we have that could be very effective for treating Parkinson’s disease, but they are just too big to cross this blood-brain barrier.
How were you able to penetrate the blood-brain barrier in this study?
For this study, the way that we get through the blood-brain barrier is, we open it with focused ultrasound and working together with our colleague, Dr. Lipsman at Sunnybrook, we used MRI to image those regions where we want to open the blood-brain barrier. We imaged people before they got the treatment and after they got the treatment, and can actually use a specific type of dye that shows us that the blood-brain barrier has opened and allowed things to enter into that very specific region of the brain. It’s a temporary opening, which is something that you want, because you don’t want bad compounds entering the brain too. Typically, to do this, we would have had to open up the skull and inject the treatment directly into the brain, which is a more invasive procedure, as you can imagine. So, to be able to see this done in real time with a patient lying in an MRI machine, without having to do open brain surgery, is remarkable.
Tell us about Pat Wilson, the first patient to be enrolled in this study.
Pat was diagnosed seven years ago at age 56 with a genetic form of Parkinson’s disease. This is actually not as common as some people think, only about 5 per cent of our Parkinson’s patients have a genetic form of the disease. Pat has something called a GBA gene mutation. All of the patients in this study have a GBA gene mutation and are otherwise healthy as we want to minimize the risk of any other medical issues arising. In Pat’s case, she also has the memory of her father living with Parkinson’s and so really was interested in doing something to help others.
It takes a special type of person to agree to take part in a safety trial, as there is no guarantee they’ll be helped in some way. It’s really about helping other people living with Parkinson’s in the future. Pat is very much wanting to advance the science and the potential understanding of this technology.

Pat Wilson (pictured with her husband, Jamie) was the first patient enrolled in the Phase I clinical trial. Pat lives with a genetic form of Parkinson’s. “If this could help somebody else, that’s great,” she says. (Photo: Courtesy Pat Wilson)
Why is personalized medicine the future of Parkinson’s treatment?
Approximately 25 years ago, we didn’t think that Parkinson’s disease was related to genes at all. But, now we know that genetics plays a very important role in Parkinson’s disease. And with that, we know that there are treatments that are probably going to be effective for people with Parkinson’s with very specific genetic abnormalities. This is what we refer to as personalized or precision medicine. This is going to be one of the very first studies where we are actually using that kind of approach. We’re selecting patients who have a known genetic abnormality and the treatment that we’re giving them is very much designed to target that genetic abnormality.
What are the next steps in this research? What should patients take away?
I think what should be taken away from this study is that we’re really at the beginning of a very exciting time in terms of discovering and developing treatments for people with Parkinson’s disease. I think this is going to be the first step of hopefully a large amount of research looking into different drugs that we could deliver in this way. Our next steps are to use this technology and this drug or other drugs and test whether we can actually have an impact on the disease progression. I am cautious but also optimistic that this will be the beginnings of what I hope will be a cure for Parkinson’s disease. And although we won’t have an immediate cure today, I really do think this is the start of laying down the foundation for our future work. The pace of research in this area can be slow and so, being involved in this kind of trial is really exciting and keeps us going.
UHN’s Parkinson’s research is supported by the Krembil Foundation and the Toronto General & Western Hospital Foundation.
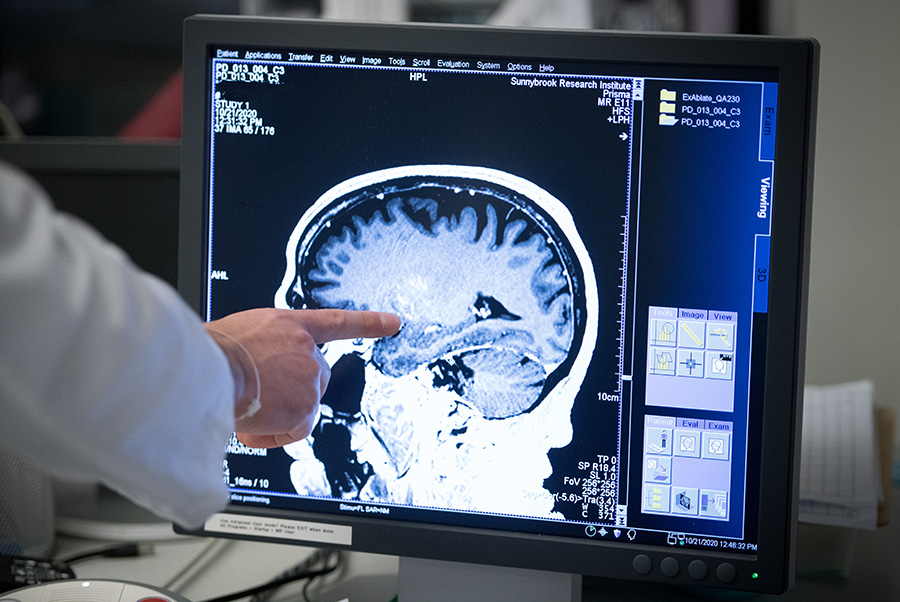
For the first time, focused ultrasound using MRI is being used to open the blood-brain barrier in patients with Parkinson’s disease. (Photo: Sunnybrook Health Sciences Centre.)


