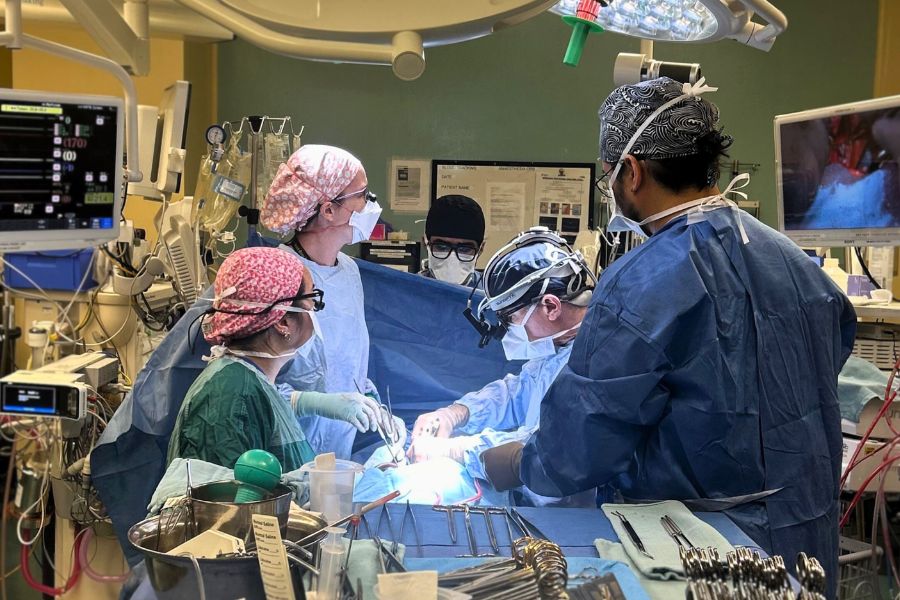
Drs. Shaf Keshavjee and Marcelo Cypel will be launching a clinical trial in May to test the viability of a new treatment for COVID-19. For their previous research accomplishments together, they were named Co-Inventors of the Year at UHN in 2019. (Photo by UHN)
With global cases recently surging past three million, novel therapies to treat COVID-19 patients are desperately needed. Presently, there are no proven interventions to prevent progression of the disease or to rapidly treat patients with COVID-19 experiencing respiratory failure.
Dr. Marcelo Cypel, thoracic surgeon and Surgical Director of the Soham & Shaila Ajmera Family Transplant Centre and Dr. Shaf Keshavjee, Surgeon-in-Chief of the Sprott Department of Surgery, are preparing to launch a clinical trial to test the effectiveness of high-dose inhaled nitric oxide in preventing the progression of COVID-19, when administered at an early stage of hospital admission. Donor funds are making it possible.
“The impact that COVID-19 can have on the individual is wide, ranging from mild signs of upper respiratory tract infection to severe pneumonia and, sadly, death,” says Dr. Cypel. “Globally, 15-18% of patients deteriorate to the point of needing mechanical ventilation to help them breathe. The probability of progression to end-stage disease is unpredictable; however, if we can prevent progression in patients with mild to moderate disease, that would lower morbidity and mortality rates and decrease the use of finite healthcare resources.”
Inhaled nitric oxide: hope for a treatment
Nitric oxide is a molecule that is produced naturally in the body and is vital to many aspects of overall health. One of its most important functions is to relax the inner muscles of the blood vessels, causing them to widen and increase circulation, allowing blood, nutrients and oxygen to travel to every part of the body effectively and efficiently.
Inhaled nitric oxide is routinely used at low doses in post-lung transplant care and for acute respiratory distress syndrome. However, some pre-clinical and early studies have pointed that nitric oxide, when given at a concentration five times higher than usual, has marked antibacterial and antiviral effects.
Importantly, high dose nitric oxide has been shown to prevent replication of SARS virus in laboratory tests. A genetic analysis revealed that current COVID-19 shares almost 80% of the same genes as the SARS virus. In humans, high dose nitric oxide has already been shown to be safe and effective against very resistant bacteria.
In this trial, scheduled to launch by May 1, high dose nitric oxide will be mixed with oxygen and delivered to patients with COVID-19 for a period of 30 minutes, three times per day, for five consecutive days from time of enrollment. The researchers believe this treatment could rapidly eliminate the virus from both upper airways and lungs in patients with COVID-19.
“If successful, the primary outcome of the trial will be a reduction in of patients requiring intubation and mechanical ventilation, with fewer patients progressing from a mild to a severe form of COVID-19,” says Dr. Cypel. “We will also be monitoring how quickly people recover, sustainability of their recovery, their health outcomes and the length of time they are admitted to hospital, compared to patients not receiving nitric oxide at other Toronto hospitals.”
UHN researchers have unique expertise and experience administering high-dose nitric oxide in a safe and reliable manner in donor lungs. As one example, our researchers have been studying the use of nitric oxide for two years in the laboratory to treat bacteria and other infections in donor lungs.
Thanks to the support of donors, phase one of this innovative trial (30 patients) is now fully funded. If the team sees positive results as the trial is rolled out over the next two to three months, they will scale up the number of participants in phase two.