Emile Faheim participated in a groundbreaking trial that allowed him to stop taking immunosuppressive medications after his liver transplant (Photo: Tim Fraser)
By Glynis Ratcliffe
When Egyptian-born Emile Faheim came to Canada in 1990 with his wife and three daughters, he thought he was perfectly healthy. But in 1994, the regional director for a fire safety company went for routine blood work and the results showed something strange: he had elevated liver enzymes in his blood. He had hepatitis C, a viral infection that can cause significant liver damage. “It was shocking, because I felt fine,” he says.
Faheim had no idea how he contracted this disease. About 75 per cent of those infected develop chronic hepatitis C, which stays in the body and progresses silently for years or even decades before finally being identified during routine blood work – exactly how Faheim’s infection was discovered.
Medications helped keep his liver inflammation under control, but then the drugs stopped working. His body began to deteriorate, with fluid accumulating first in his midsection and later in his lungs, requiring regular trips to the hospital. In 2009, Faheim was placed on a list to receive a liver transplant. In 2013, he received the call from the Ajmera Transplant Centre at University Health Network (UHN): they had found a liver for him. It was in his body the next day. “It was a miracle, truly,” Faheim says.
As excited as he was to have a new liver, Faheim wasn’t out of the woods yet; his body needed to accept the new organ as its own and he had to ensure his hepatitis C wouldn’t get worse. For the latter, he took a drug called Sovaldi, a “magic cure,” he says, that was first trialled by Dr. Jordan Feld, Co-Director of the Schwartz Reisman Liver Research Centre and the R. Phelan Chair in Translational Liver Research at UHN. After three months of treatment the hepatitis C disappeared.

Having his body accept his new liver was a bigger challenge. One thing all transplant patients have in common is the potential for the body to reject the new organ. Most have to take strong medications for the rest of their lives to prevent the body from attacking its new addition. The medications can come with serious side effects. For Faheim, they caused his blood pressure to rise and his blood sugar to become unstable. This resulted in him having to take more medications.
Thankfully, Faheim went on to participate in a trial that may make onerous post-operative drug regimens a thing of the past.
Learning not to attack
When the immune system encounters something it doesn’t recognize in the body, it mounts a battle to destroy that foreign agent – whether it’s an organ, a virus or even a splinter. It’s how our bodies fight disease, but it becomes a problem when the foreign object being attacked was placed there to save that person’s life.
The immunosuppressive drugs transplant recipients take to prevent the body from rejecting the new organ are toxic medications that don’t always work long term, says Dr. Stephen Juvet, Deputy Research Director of the Ajmera Transplant Centre.
With anti-rejection medication, patients can experience high blood pressure, fatigue, decreased blood cell counts, intestinal upset and high cholesterol, among other side effects. Scientists at the Centre are focused on finding ways to help the body better accept organs.
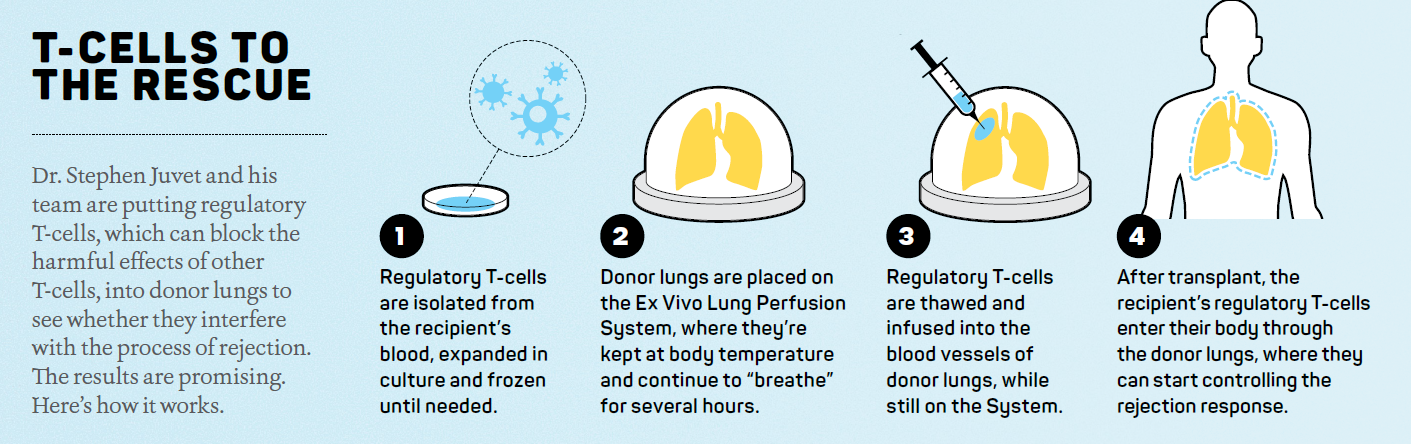
Dr. Juvet is just one of the clinician-scientists at the Ajmera Transplant Centre who are on the case. He oversees groundbreaking work on immune tolerance, working with all organ types. His other main focus is understanding the mechanisms of chronic lung allograft rejection, or why lungs can continue to be rejected well after they are transplanted into a patient. He wants to find ways physicians can intervene earlier in the chronic rejection process, which is more of a long-term issue. “People think of a transplant as sort of a cure, but it’s not that,” says Dr. Juvet, noting that a person’s new organs don’t last forever and other treatment options may be required.
Lungs have the highest rate of rejection, with a 55 per cent patient survival rate at five years post-transplant. Dr. Juvet believes some instances of rejection have to do with the early inflammation that happens at the time of surgery, what’s known as ischemia reperfusion injury. This happens when an organ is removed from the donor and put on ice or in cold storage during transportation. As the organ is rewarmed and filled with the recipient’s blood, the resulting inflammation may contribute to acute (within the first year) or chronic (long-term) rejection. But another reason is because the lungs are constantly exposed, through the airways, to inflammation triggers like air pollution in the external environment and acid reflux. In both cases, inflammation is the key driver of transplant rejection.
“The central idea of tolerance is about getting T-cells, a type of white blood cell that is part of the immune system, to not respond, despite seeing this foreign organ in the body,” Dr. Juvet explains.
A LITMUS test
To help get people off immunosuppressants, Dr. Juvet is overseeing an ongoing clinical trial for liver transplant patients called the Liver Immune Tolerance Marker Utilization Study (LITMUS).
The trial, which ultimately freed Faheim from the drug regimen he was taking post-transplant, began in 2015 and was created by principal researcher Dr. Gary Levy, former Director of what is now the Ajmera Transplant Centre, who worked on immune tolerance research for years at UHN. Faheim was one of 60 patients the LITMUS research team approached to see if he would be willing to try a controlled weaning off his anti-rejection drugs, after a blood test confirmed the presence of a tolerance biomarker that Dr. Levy had previously identified. That biomarker, which could be a game-changer but needs more study, indicates which transplant patients may be able to stop taking those drugs altogether.

Another scientist pursuing new immune tolerance strategies is Dr. Sarah Crome, a fundamental research scientist with the Ajmera Transplant Centre. Fundamental science aims to understand the underlying basis of disease or health. “My lab is focused on defining how your immune system decides when and when not to attack,” explains Dr. Crome. “So, what are the cellular and molecular processes that control immune response?”
A new type of partnership
Dr. Crome uses patient samples wherever possible, because it brings her closer to understanding the mechanisms of disease. She works alongside UHN clinicians for this reason. “We sit together, and they’ll say this is the clinical presentation, this is the problem we’re having, this is how the disease is manifesting,” she says. “And then I take that to the lab and ask, ‘Is there something we can find that explains what is observed clinically?’”
This is what led Dr. Crome to look closer at the cells in tissue biopsies from different organs. It turns out that the cells in organ tissues are very different from those in circulation in the blood, with specialized functions that may affect immune tolerance. “If we can identify the cells that are responsible for starting that rejection reaction,” she explains, “or understand specific molecules that trigger rejection or prevent it, we could devise new immunotherapy approaches that are more specific and more targeted than general immunosuppression.”
The hope is that more transplant recipients will be identified as having higher tolerance biomarkers, moving beyond liver transplant patients like Faheim to potentially include other organs. It would certainly improve many people’s quality of life.
Read more in the Ajmera Transplant Centre magazine.